Description
Designed for maximum comfort. Sciessent, a U.S.-based global provider of antimicrobial solutions, has incorporated its Agion® treatment into the fabric. The Agion treatment incorporated into masks is modeled after the FDA, EU and Canadian cleared Nexera surgical respirator, that relies on Agion Antimicrobial performance to make FDA cleared claims to inactivate viruses by 99.99% in 5 minutes and kill 99.99% of bacteria in one hour. Learn more about Sciessent.
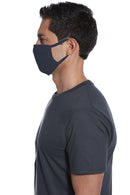
This three-ply, 100% cotton jersey face mask covers the wearer's nose and mouth. Elastic stretch binding ear loops help hold it in place.
- Three-ply 4-ounce, 100% cotton jersey
- Fabric touching skin: 100% cotton
- Mask contains silver and copper Three evenly-spaced pleats on each side
- Non-returnable
- May be used when FDA-cleared masks are unavailable
- Not recommended for use in a surgical setting or where significant exposure to liquid bodily or other hazardous fluids may be expected
- The product has been authorized by FDA under an EUA for use by HCP as PPE to help prevent the spread of infection or illness in healthcare settings and by the general public to help slow the spread of the virus during the COVID-19 pandemic.
- This product is authorized only for the duration of the declaration that circumstances exist justifying the authorization of the emergency use of medical devices, including alternative products used as medical devices, during the COVID-19 outbreak, under section 564(b)(1) of the Act, 21 U.S.C. § 360bbb-3(b)(1) unless the authorization is terminated or revoked sooner.
How to Customize Safety Vest?
Customer Reviews
Payment & Security
Your payment information is processed securely. We do not store credit card details nor have access to your credit card information.